Epoxy plastics’ general chemical and physical properties
An epoxy resin is defined as a molecule with more than one epoxy group, which can be hardened into a usable plastic. The epoxy group, which is also called the glycidyl group, has through its characteristic appearance given the name to epoxy.
What one sees is an oxygen atom on the outside of the carbon chain. Epi means “on the outside of” and the second part of the word comes from oxygen.
There are two spellings, namely epoxi and epoxy. The first comes from the oxygen’s bond with the carbon chain being called an oxide. Epoxy resin is manufactured from simple basic chemicals that are readily available.
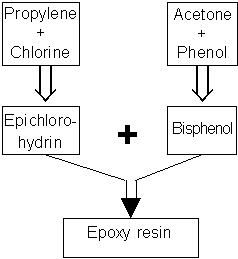
With the help of chemical formulas, the last stage out is as follows:
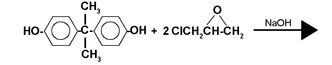 Bisphenol A + Epichlorohydrin
Bisphenol A + Epichlorohydrin
By varying the relationship between bisphenol A and epichlorohydrin, various molecular weights are obtained for the completed epoxy resin. The lowest molecular weight an epoxy resin of the DGEBA type can have is 340, but if two elements together can form different molecular weights when they react, the epoxy resin will contain a mixture of epoxy molecules of varying lengths. One therefore does not refer to the epoxy resins’ molecular weight, but rather to their mean molecular weight.
Epoxy resin with a mean molecular weight of over 700 is called high molecular, and epoxy resin with a mean molecular weight of under 700 low molecular. Epoxy resins can be allergens, and it is the molecular weight that determines how great the risk. The higher molecular weight, the lower the probability for allergies.
In the formula for epoxy resin, the letter “n” is seen after the brace. If n=0, i.e. that which is inside the brace does not exist, we then have the shortest epoxy molecule with a molecular weight of 340. It has the highest reactivity and thus also constitutes the greatest allergy risk. If n=1, the molecular weight is 624, for n=2 it is 908, etc.
For each time n increases by 1, the molecular weight increases by 284. In a low molecular epoxy resin with the mean molecular weight of 380, the distribution is approximately 88% n=0, 10% n=1 and 2% n=2. A pure epoxy resin with n=2 is not an allergen, but if we look at a commercial epoxy resin with the molecular weight of 1080, the distribution is approximately 20% n=0, 15% n=1, 15% n=2 and 50% n=3, 4 and 5. This means the even a high molecular epoxy resin can be an allergen.
A low molecular epoxy resin with a mean molecular weight of 380 is fluid at room temperature, while an epoxy resin with a mean molecular weight of 1000 is solid at room temperature. The molecular weight determines what the epoxy resin can be used for.
The low molecular can be handled without solvent additives, which evaporate and are therefore used for casting, thick coatings, gap-filling glues, etc.
The high molecular epoxy resins must as a rule be dissolved in organic solvents to be manageable, which limits usage to paints and lacquers.
To convert epoxy resin to epoxy plastic, a reaction with a suitable substance is required. Such a substance in this context is called a hardener.
Examples of substance groups that function as epoxy hardeners are: amines, amides, acid anhydride’s, imidazoles, boron trifluoride complexes, phenols, mercaptans and metal oxides.
For hardening at room temperature, amines and amides are primarily used, and to a certain extent mercaptans. The other hardener types generally require temperatures above +150°C to react with the epoxy. From this point forward, only amines and amide hardeners will be further described.
Amines are substances that are closely related to ammonia (NH3). Depending on how many hydrogen atoms that are replaced by alkyl groups, primary amines NH2-R, secondary amines NH-R1 or tertiary amines N-R2 arise. The total number of amino groups determines if the amine is a monoamine (NH2-R), a diamine (NH2-R-NH2) or a polyamine (NH2-R-NH-R-NH-R-NH2).
Furthermore, the amines are divided into aliphatic, i.e. a straight carbon chain, cycloaliphatic with a ring-shaped carbon chain and finally aromatic where the amino group is bonded to a benzene ring.
As hardener for the epoxy resin, primarily diamines and polyamines are used.
The primary amino group NH2 contains as seen, two hydrogen atoms and one nitrogen atom. It is the hydrogen that constitutes the reactive part, and the reaction occurs with the oxygen in the epoxy group.
With somewhat simplified chemical formulas, it looks like this:
 Primary amine + Epoxy group gives Secondary amino group
Primary amine + Epoxy group gives Secondary amino group
In the first reaction phase, one of the amine’s hydrogen atoms reacts with the epoxy group’s oxygen, causing the formation of a hydroxyl group (OH–) at the same time as the primary amine is reduced to a secondary amine. The reaction continues:

The secondary amine reacts with yet another epoxy group and the reaction is complete.
An epoxy molecule normally contains two epoxy groups, and one primary diamine has four reactive hydrogen atoms. A schematic of the epoxy plastic then looks like this:

The epoxy plastic molecule is of course three-dimensional in reality.
Examples of amines that are used as hardeners for epoxy resins
Aliphatic amines
- Diethylenetriamine
- Triethylenetetramine
- Aminoethyl piperazine
- Trimethyl hexamethylenediamine
Cycloaliphatic amines
- Isophorondiamine
- Diamino-dicyclohexylmethane
Aromatic amines
- Diaminodiphenylmethane
- m-Phenylendiamine
It is very common to pre-react the amines with a certain portion of epoxy resin. The purpose of this is to attain a hardener that is less fluid than the pure amine and that has a somewhat higher reactivity.
This type of hardener is called amine adducts, and reacts largely in the same way as previously shown.
The next large group of hardeners is the amides, or more correctly expressed, the polyaminoamides. An amide is formed when a polyamine reacts with a fatty acid.

This generally applies to all polyamides, even nylon. By varying the relationship between fatty acid and amine, one can decide if the polyamide will be acid-terminated (of the type nylon) or amine-terminated, i.e. which end groups the polyamide will have. Only amine-terminated polyamides can be used as hardeners for epoxy resin. As fatty acid, most often used is tall fatty acid, linolic acid or olein. The fatty acids use either monomer (one carboxylic group) or dimer (two carboxylic groups). As polyamines, diethylenetriamine, triethylenetetramine and tetraethylenepentamine are used. The hydrogen in the amide group (CONH) is not reactive, but it is rather the hydrogen in the primary amino groups at the polyamide’s ends and the secondary amino groups that are derived from the polyamine, which are reactive with the epoxy resin in the same way as previously described.
Both amine adducts and polyamides can be made water soluble. Such solutions have the capacity to emulsify low molecular epoxy resin, which in turn provides the possibility to manufacture water soluble epoxy paints.
The reaction between an epoxy resin and a hardener is an irreversible poly-addition, i.e. no by-products are formed, and the epoxy plastic cannot be decomposed into epoxy resin and hardener. The reaction is exothermic, which means that heat is released. Depending on the type of hardener one uses, one can achieve very large differences in reaction speeds. This has significant practical importance when working with epoxy. The time it takes to consume a mixture of epoxy resin and hardener is called potlife. Depending on the mixture’s reactivity, potlife can vary from a few seconds to several years.
Potlife can be determined in several ways.
One method is to temper the epoxy resin and hardener to +20°C. Thereafter an amount of 100 grams is mixed in a plastic cup. The time for the mixture’s temperature to reach +50°C is set as the mixture’s potlife. For systems with low reactivity (long potlife), one most often chooses to measure the viscosity or consistency, and measure the time until the initial viscosity is doubled.
Both of these methods are unusable for aqueous emulsified or aqueous dispersed systems. In this case, one instead performs a lay-up test and measures the time to gloss reduction.
Most chemical reactions follow Arrhenius’ law, which states that the reaction speed doubles for each tenth of a degree the temperature is increased. This means that a reaction is twice as fast at +30°C as at +20°C.
The larger the amount that is mixed together, the greater the amount of exothermic heat that is created. There is not enough time for this heat to be dissipated through the mixture vessel’s surface, so instead it heats the mixture. As the temperature increases, the reaction speed also increases, which entails that the potlife is shorter, the larger the amount that is mixed.

Example of potlife for 100 and 500 grams of the same epoxy resin/hardener mixture
Potlife for an epoxy system consequently provides certain information to the user on working time after mixture, but one must consider the amount of material mixed, and the material’s initial temperature. The epoxy’s hardening time is defined as the time from the epoxy being applied until the formed epoxy plastic has achieved its final properties pertaining to strength and chemical resistance.
For epoxy that is applied in thin layers, the exothermic heat will not increase the temperature in the layer to any significant degree, but instead, the epoxy quickly assumes the temperature of the substrate. The reaction between the epoxy resin and hardener then goes relatively quick at the beginning because of the large availability of reactive molecules, and because the mobility of the molecules is high as long as the viscosity is low.
As complete epoxy molecules are formed, the number of reactive molecules is reduced at the same time as the viscosity increases. The reaction speed gradually slows.
As a rule of thumb, room temperature hardened epoxy needs about 7 days at +20°C to attain maximum properties, but already after 24 hours, one can have attained about 70–80% of the final properties.
Arrhenius’ law naturally applies even if the reaction occurs at a constant temperature. This means that if the substrate is at +10°C, it takes about 14 days to attain final properties.

Example of epoxy’s hardening process at constant temperature.
Epoxy plastic’s characteristic basic properties
Currently more than 50 different substances fulfil the definition for an epoxy resin. If one also adds that there are several hundred different hardeners, it is easy to understand that epoxy plastic’s properties can be modified to satisfy the most varied requirements. Nevertheless, certain basic properties are always present.
Adhesion
One of epoxy plastic’s most characteristic properties is the capacity to adhere to most substrates. The reason for this is the presence of polar hydroxyl groups, and the ether bonds. The negligible shrinkage also entails that contact between the epoxy plastic and the substrate is not disturbed by tensions. The epoxy plastic’s surface tension is most often under the critical surface energy for most materials. This is one of the requirements for adhesion to be achieved.
Mechanical strength
No other hard plastic can display as high mechanical strength as correctly formulated epoxy plastic. Again, it is largely because of the minimal shrinkage that built-in tensions are avoided. The tensile strength can exceed 80 MPa.
Chemical resistance
Thanks to the possibility of varying the epoxy plastics’ properties, one can make epoxy plastic resistant to most chemicals. In general, epoxy plastic is very resistant to alkali, which is of importance in surface-treating concrete.
Diffusion density
Epoxy generally has relatively high vapour transmission resistance, but with a special technique, epoxy plastic can be made open to diffusion. Epoxy that is open to diffusion can be applied on, for example, wet concrete and provide adhesion higher than the concrete’s tensile strength.
Water tightness
The epoxy plastics are to be considered as watertight and they are often used to protect against water.
Electrical insulation capacity
Epoxy plastics are excellent electrical insulators. Volume resistivity is normally 1015Ohm·cm. This in combination with high moisture resistance and chemical resistance makes the epoxy suitable for both the manufacture of electronics components and the embedment of transformers.
Shrinkage
The epoxy plastics have very slight shrinkage during hardening. This is because the epoxy molecule has a rather small reorientation during the hardening process compared with, for example, polyester and methylmetacrylate.
Heat resistance
When it comes to heat resistance, room temperature hardened epoxy plastic differs very little from heat hardened. One often specifies heat resistance with HDT (Heat Deflection Temperature) or Tg (Glass transition temperature).
At HDT, the mechanical strength declines quickly. Room temperature hardened epoxy seldom attains HDT above 70°C, while heat hardened can reach 250°C.
Modifiable
Perhaps the chief property of epoxy is the nearly unlimited capability to modify the final properties of the epoxy plastic to meet special requirements. It is primarily the hardener that influences the plastic’s properties, but as presented in the next chapter, there are many other substances that influence epoxy products.
Stability in light
Epoxy plastics based on aromatic epoxy resins are sensitive to light in the UV range. Direct radiation with ultraviolet light quickly causes yellowing. Even normal sunlight contains enough ultraviolet radiation for yellowing to occur. Most resistant are aliphatic epoxy resins with anhydride or amine hardener.
Modification of epoxy resins
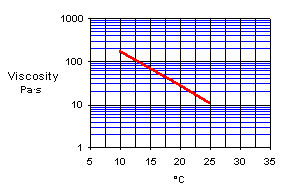
Viscosity at 25°C of an unmodified low molecular epoxy resin of the DGEBA type, is about 10 Pa·s, at 20°C about 24 Pa·s and at 15°C about 68 Pa·s.
It is easy to understand that an unmodified epoxy resin cannot always be used outdoors or where the temperature is low. The first reason for modification is thus viscosity reduction to a suitable working consistency. Several options are available here.
First, we have non-reactive diluents, which refers to such substances that can be mixed with the epoxy resin but that do not participate in the reaction between the epoxy and hardener. Included here are the ordinary solvents such as xylene, toluole, glycol ethers, ketones, lower alcohols, etc.
Other non-reactive diluents include a large number of substances with sufficiently low vapour pressure that they do not evaporate from the hardened plastic under normal conditions. Examples of these are benzyl alcohol and coumarone resins. The substances that are not chemically bonded in the epoxy plastic molecule are to be imagined as deposited between the plastic molecular chains.
In this group are the “ordinary” solvents that have the strongest influence on viscosity, but which often entail a dangerous path.
Epoxy resin has very high solvent retention, i.e. the capacity to retain solvents. This means that it will take a long time before the solvent has evaporated. In a 1 mm thick layer, solvent residue can be detected after several months at room temperature.
Damage that can arise from solvents is usually blistering, either through high heating or because of osmosis when moisture occurs in concrete. Another reason to avoid solvents is the shrinkage effect that can result in layers detaching from weak substrates.
The diluents that under normal conditions do not evaporate affect the epoxy plastic’s properties in other ways than viscosity reduction. Positive changes can be flexibility, improved resistance to water and salt solutions because of hydrophobing, improved reactivity, in part because the mobility of the epoxy and the hardener molecule is better at lower viscosity, and in part because of the catalytic effect from hydroxyl groups in, for example, benzyl alcohol.
Negative changes are degraded heat resistance and larger thermal expansion.
At higher temperatures, volatility can be substantial with several of these substances. This can result in shrinkage with cracking and subsequent reduced adhesion. Another aspect that sometimes must be considered is the compatibility with bitumen. A minority of the non-reactive diluents do not bleed in bitumen.
Furthermore, many diluents in this group are esters that can saponify upon contact with concrete under the influence of water.
Reactive diluent (thinners)
These substances have, as the name implies, the capacity to react with the epoxy resin or hardener, so that in this way, bond in the plastic molecule.
The most used types are those that contain one or more epoxy groups. There is a very large group of such substances that all can give the final plastic different properties.
What characterizes these substances is that they cannot migrate, evaporate or be extracted from the epoxy plastic. Heat resistance declines somewhat in comparison to unmodified epoxy, but not at all as much as in the case of the non-reactive diluent. A valuable property of the reactive diluent is that they reduce the surface tension of the epoxy resin, which in turn can improve wetting capabilities and thus adhesion. Depending on the reactive diluent composition, both flexibility and chemical resistance can be affected.
Other substances used to modify the epoxy plastic’s properties are, for example, high molecular isocyanates, which via the hardener, can react with the epoxy resin. The result is a plastic with rubber-elastic properties, which are retained down to about -40°C.
Such modification changes the epoxy plastic’s fundamental properties.
Heat resistance and chemical resistance decline with increased elasticity. At the same time, the viscosity increases because the isocyanates in themselves are very highly viscous. This means that further modification is necessary to reduce viscosity.
The next group of modification substances is pigments and fillers.
Pigment is used to colour the epoxy material, and the filler to increase the mechanical strength and to reduce costs.
The pigments used are most often metal oxides such as titanium dioxide, iron oxide and chromium oxide.
As a rule, fillers are finely ground minerals and quartz sand. It is important that both pigments and fillers are properly dispersed in the epoxy binding agent.
In addition to the listed modification substances, there are a number of aids to affect such things as rheology, i.e. consistency, flow, air bubbles and adhesion.
It is easy to understand that an epoxy product has a relatively complex composition, where the included components shall interact for the results to be as intended.